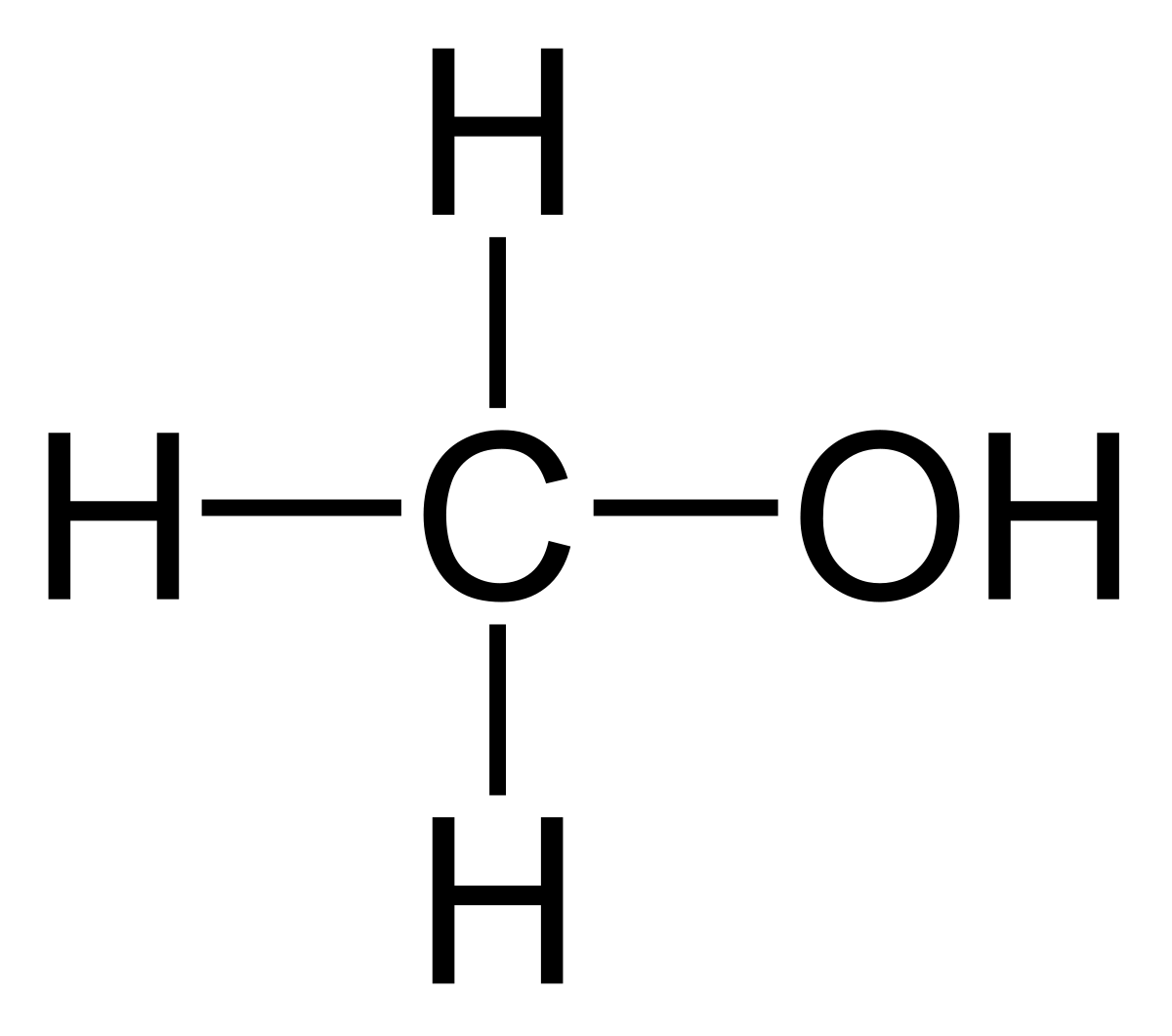
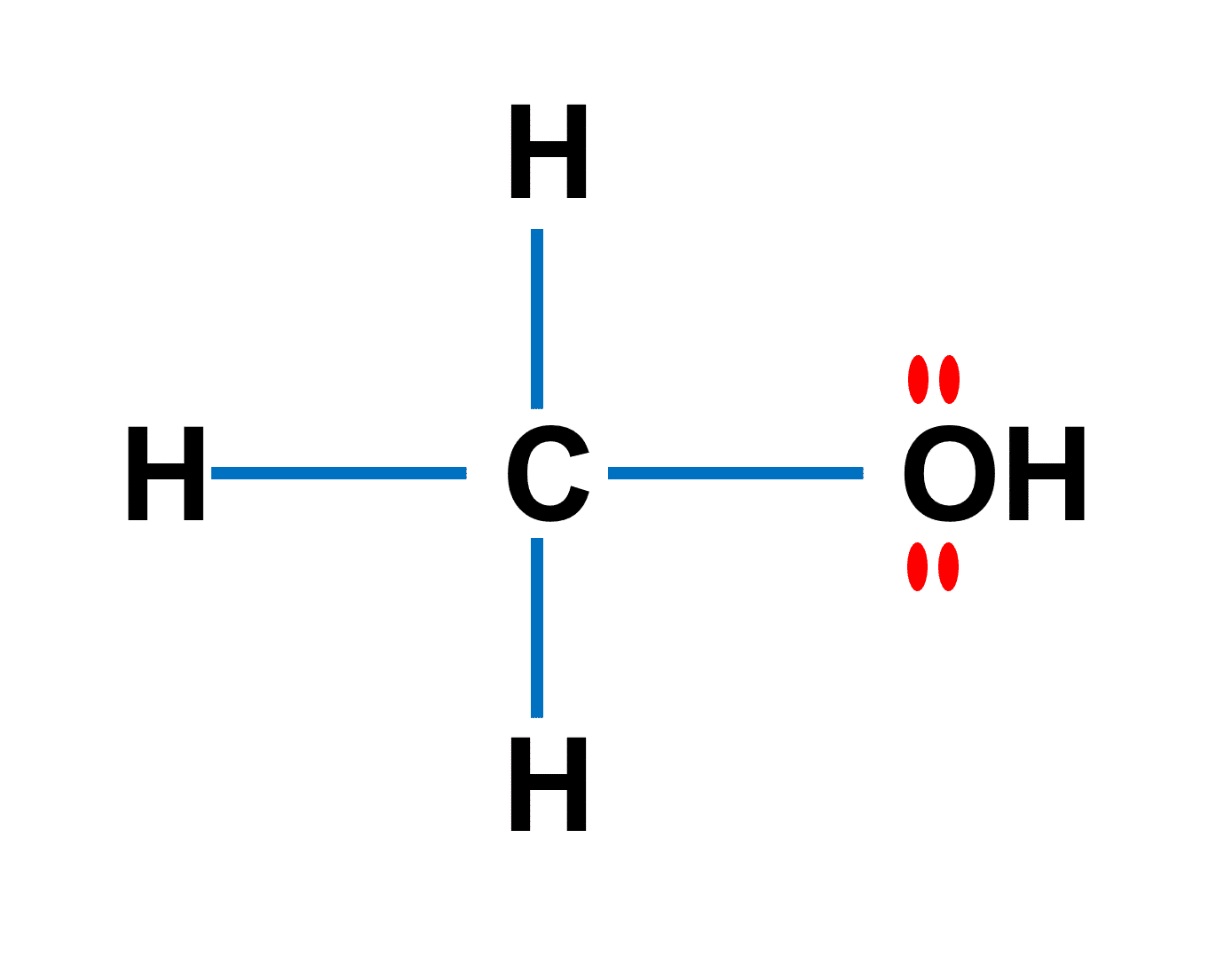
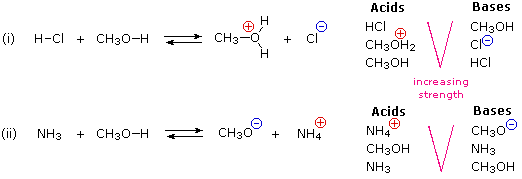
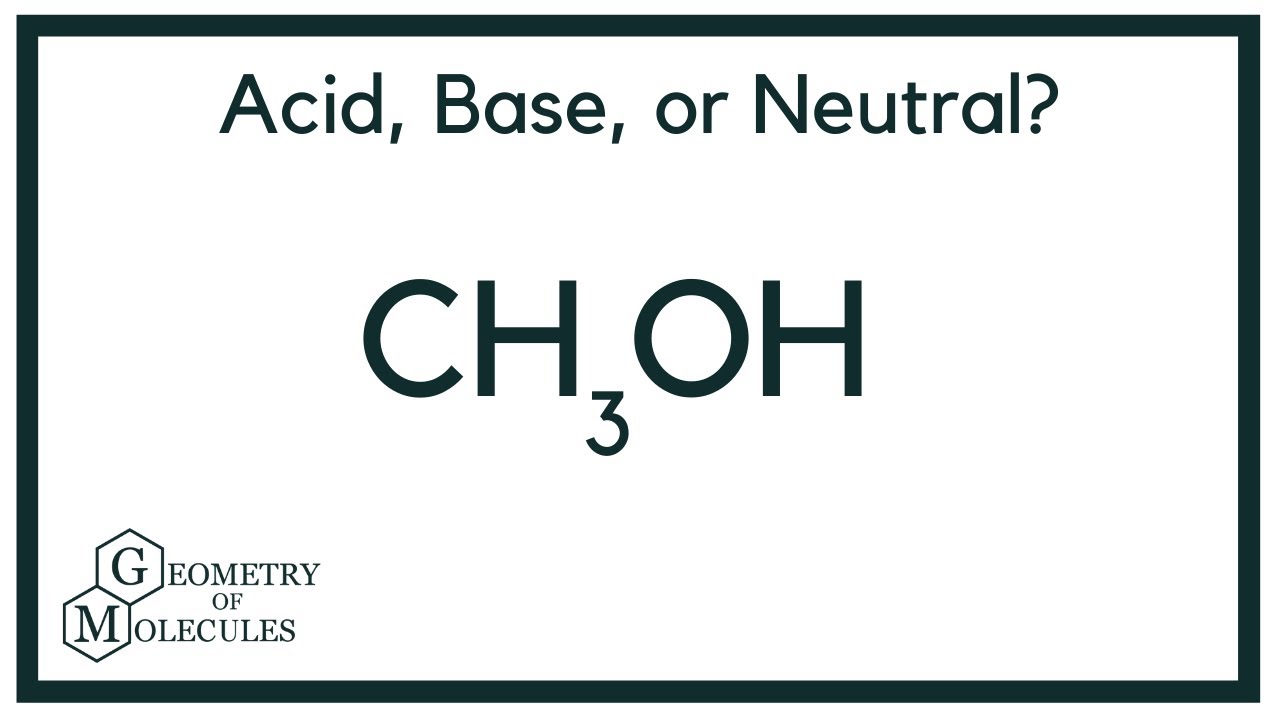
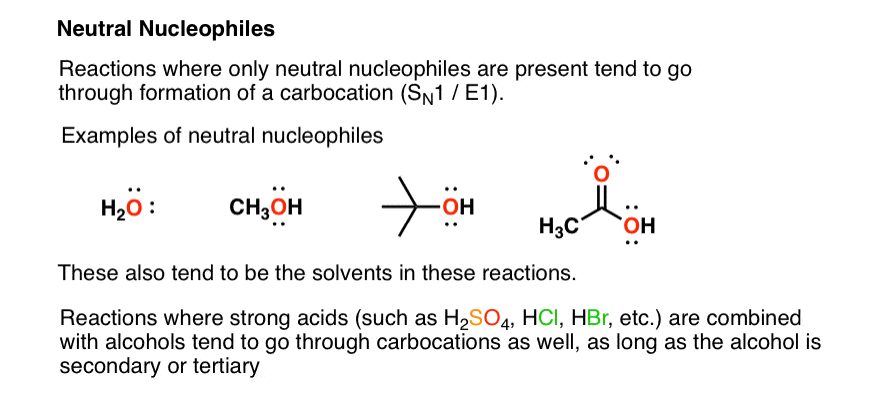
Can methanol be classified as an Arrhenius acid? To my knowledge it does not want the H from the O--H bond to dissociate : r/Mcat

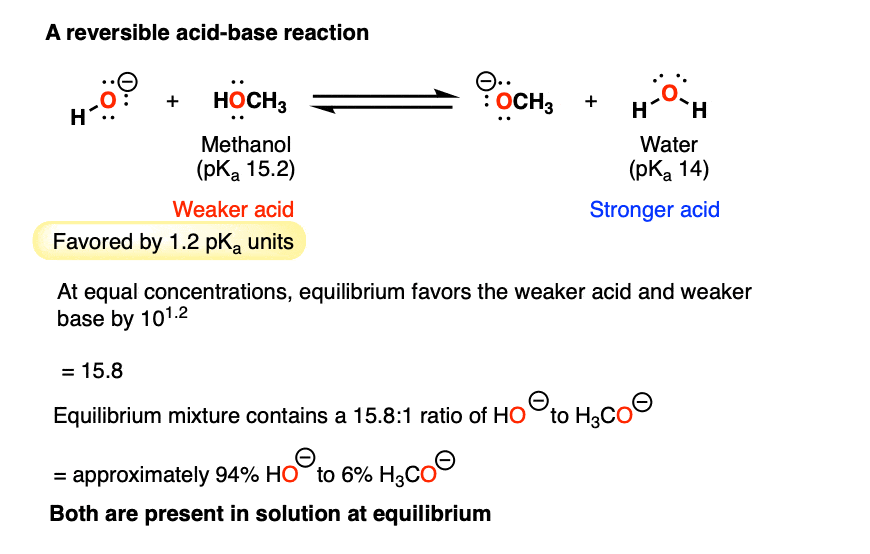
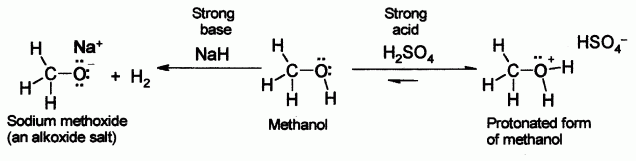
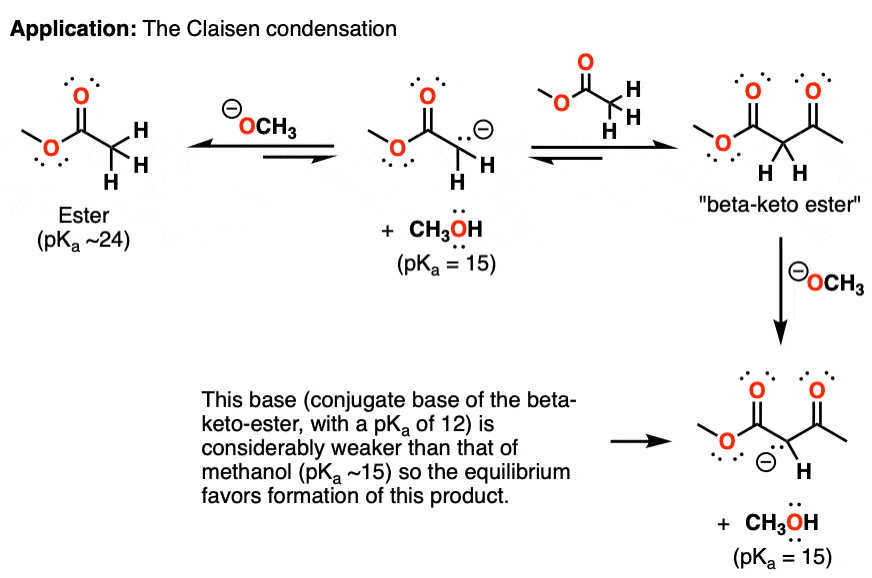
SOLVED: Match each substance in the following equation as Acid (A), Base (B), Conjugate Acid (CA), or Conjugate Base (CB) CH3OH NH2 CH3O" NH3 Conjugate Acid CHzOH Acid NH2 Conjugate Base CH3o-
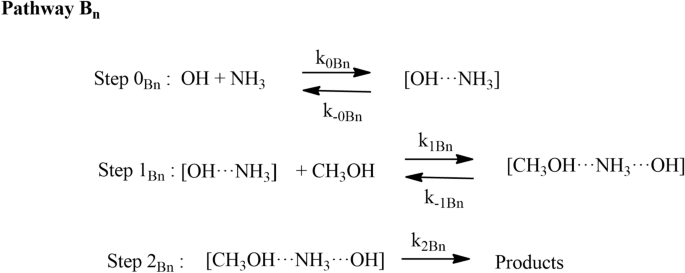
Effect of ammonia and water molecule on OH + CH3OH reaction under tropospheric condition | Scientific Reports